| << Chapter < Page | Chapter >> Page > |
Another example of energy conversion occurs in a solar cell. Sunlight impinging on a solar cell (see [link] ) produces electricity, which in turn can be used to run an electric motor. Energy is converted from the primary source of solar energy into electrical energy and then into mechanical energy.

| Object/phenomenon | Energy in joules |
|---|---|
| Big Bang | |
| Energy released in a supernova | |
| Fusion of all the hydrogen in Earth’s oceans | |
| Annual world energy use | |
| Large fusion bomb (9 megaton) | |
| 1 kg hydrogen (fusion to helium) | |
| 1 kg uranium (nuclear fission) | |
| Hiroshima-size fission bomb (10 kiloton) | |
| 90,000-ton aircraft carrier at 30 knots | |
| 1 barrel crude oil | |
| 1 ton TNT | |
| 1 gallon of gasoline | |
| Daily home electricity use (developed countries) | |
| Daily adult food intake (recommended) | |
| 1000-kg car at 90 km/h | |
| 1 g fat (9.3 kcal) | |
| ATP hydrolysis reaction | |
| 1 g carbohydrate (4.1 kcal) | |
| 1 g protein (4.1 kcal) | |
| Tennis ball at 100 km/h | |
| Mosquito | |
| Single electron in a TV tube beam | |
| Energy to break one DNA strand |
Even though energy is conserved in an energy conversion process, the output of useful energy or work will be less than the energy input. The efficiency of an energy conversion process is defined as
[link] lists some efficiencies of mechanical devices and human activities. In a coal-fired power plant, for example, about 40% of the chemical energy in the coal becomes useful electrical energy. The other 60% transforms into other (perhaps less useful) energy forms, such as thermal energy, which is then released to the environment through combustion gases and cooling towers.
| Activity/device | Efficiency (%) Representative values |
|---|---|
| Cycling and climbing | 20 |
| Swimming, surface | 2 |
| Swimming, submerged | 4 |
| Shoveling | 3 |
| Weightlifting | 9 |
| Steam engine | 17 |
| Gasoline engine | 30 |
| Diesel engine | 35 |
| Nuclear power plant | 35 |
| Coal power plant | 42 |
| Electric motor | 98 |
| Compact fluorescent light | 20 |
| Gas heater (residential) | 90 |
| Solar cell | 10 |
A realistic mass and spring laboratory. Hang masses from springs and adjust the spring stiffness and damping. You can even slow time. Transport the lab to different planets. A chart shows the kinetic, potential, and thermal energies for each spring.

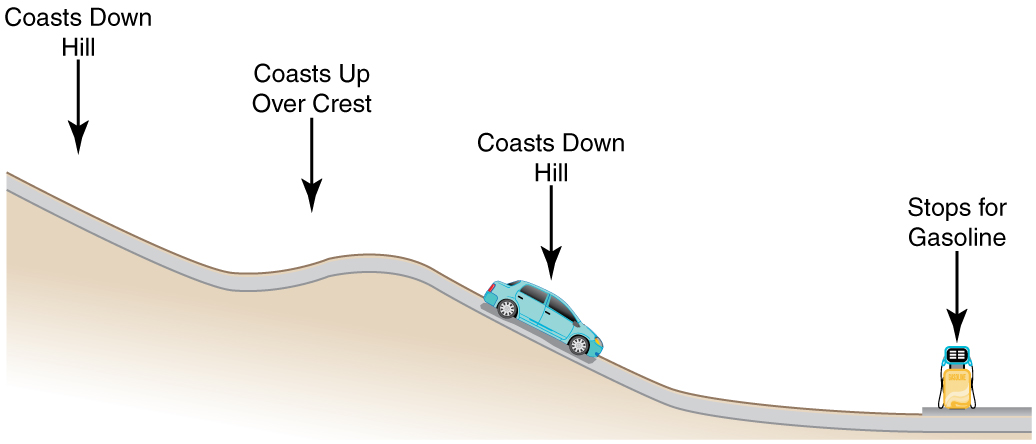
Consider the following scenario. A car for which friction is not negligible accelerates from rest down a hill, running out of gasoline after a short distance. The driver lets the car coast farther down the hill, then up and over a small crest. He then coasts down that hill into a gas station, where he brakes to a stop and fills the tank with gasoline. Identify the forms of energy the car has, and how they are changed and transferred in this series of events. (See [link] .)
Describe the energy transfers and transformations for a javelin, starting from the point at which an athlete picks up the javelin and ending when the javelin is stuck into the ground after being thrown.
Do devices with efficiencies of less than one violate the law of conservation of energy? Explain.
List four different forms or types of energy. Give one example of a conversion from each of these forms to another form.
List the energy conversions that occur when riding a bicycle.
Using values from [link] , how many DNA molecules could be broken by the energy carried by a single electron in the beam of an old-fashioned TV tube? (These electrons were not dangerous in themselves, but they did create dangerous x rays. Later model tube TVs had shielding that absorbed x rays before they escaped and exposed viewers.)
Using energy considerations and assuming negligible air resistance, show that a rock thrown from a bridge 20.0 m above water with an initial speed of 15.0 m/s strikes the water with a speed of 24.8 m/s independent of the direction thrown.
Equating and , we obtain
If the energy in fusion bombs were used to supply the energy needs of the world, how many of the 9-megaton variety would be needed for a year’s supply of energy (using data from [link] )? This is not as far-fetched as it may sound—there are thousands of nuclear bombs, and their energy can be trapped in underground explosions and converted to electricity, as natural geothermal energy is.
(a) Use of hydrogen fusion to supply energy is a dream that may be realized in the next century. Fusion would be a relatively clean and almost limitless supply of energy, as can be seen from [link] . To illustrate this, calculate how many years the present energy needs of the world could be supplied by one millionth of the oceans’ hydrogen fusion energy. (b) How does this time compare with historically significant events, such as the duration of stable economic systems?
(a)
(b) This is much, much longer than human time scales.

Notification Switch
Would you like to follow the 'College physics' conversation and receive update notifications?