| << Chapter < Page | Chapter >> Page > |
In other cases, the electrostatic attractions between the ions in a crystal are so large, or the ion-dipole attractive forces between the ions and water molecules are so weak, that the increase in disorder cannot compensate for the energy required to separate the ions, and the crystal is insoluble. Such is the case for compounds such as calcium carbonate (limestone), calcium phosphate (the inorganic component of bone), and iron oxide (rust).
Pure water is an extremely poor conductor of electricity because it is only very slightly ionized—only about two out of every 1 billion molecules ionize at 25 °C. Water ionizes when one molecule of water gives up a proton to another molecule of water, yielding hydronium and hydroxide ions.
In some cases, we find that solutions prepared from covalent compounds conduct electricity because the solute molecules react chemically with the solvent to produce ions. For example, pure hydrogen chloride is a gas consisting of covalent HCl molecules. This gas contains no ions. However, when we dissolve hydrogen chloride in water, we find that the solution is a very good conductor. The water molecules play an essential part in forming ions: Solutions of hydrogen chloride in many other solvents, such as benzene, do not conduct electricity and do not contain ions.
Hydrogen chloride is an acid , and so its molecules react with water, transferring H + ions to form hydronium ions (H 3 O + ) and chloride ions (Cl − ):
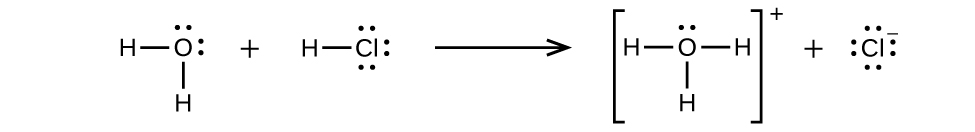
This reaction is essentially 100% complete for HCl (i.e., it is a strong acid and, consequently, a strong electrolyte). Likewise, weak acids and bases that only react partially generate relatively low concentrations of ions when dissolved in water and are classified as weak electrolytes. The reader may wish to review the discussion of strong and weak acids provided in the earlier chapter of this text on reaction classes and stoichiometry.
Substances that dissolve in water to yield ions are called electrolytes. Electrolytes may be covalent compounds that chemically react with water to produce ions (for example, acids and bases), or they may be ionic compounds that dissociate to yield their constituent cations and anions, when dissolved. Dissolution of an ionic compound is facilitated by ion-dipole attractions between the ions of the compound and the polar water molecules. Soluble ionic substances and strong acids ionize completely and are strong electrolytes, while weak acids and bases ionize to only a small extent and are weak electrolytes. Nonelectrolytes are substances that do not produce ions when dissolved in water.
Explain why the ions Na + and Cl − are strongly solvated in water but not in hexane, a solvent composed of nonpolar molecules.
Crystals of NaCl dissolve in water, a polar liquid with a very large dipole moment, and the individual ions become strongly solvated. Hexane is a nonpolar liquid with a dipole moment of zero and, therefore, does not significantly interact with the ions of the NaCl crystals.
Explain why solutions of HBr in benzene (a nonpolar solvent) are nonconductive, while solutions in water (a polar solvent) are conductive.
Consider the solutions presented:
(a) Which of the following sketches best represents the ions in a solution of Fe(NO 3 ) 3 ( aq )?
3_img.jpg)
(b) Write a balanced chemical equation showing the products of the dissolution of Fe(NO 3 ) 3 .
(a) Fe(NO 3 ) 3 is a strong electrolyte, thus it should completely dissociate into Fe 3+ and ions. Therefore, (z) best represents the solution. (b)
Compare the processes that occur when methanol (CH 3 OH), hydrogen chloride (HCl), and sodium hydroxide (NaOH) dissolve in water. Write equations and prepare sketches showing the form in which each of these compounds is present in its respective solution.
What is the expected electrical conductivity of the following solutions?
(a) NaOH( aq )
(b) HCl( aq )
(c) C 6 H 12 O 6 ( aq ) (glucose)
(d) NH 3 ( l )
(a) high conductivity (solute is an ionic compound that will dissociate when dissolved); (b) high conductivity (solute is a strong acid and will ionize completely when dissolved); (c) nonconductive (solute is a covalent compound, neither acid nor base, unreactive towards water); (d) low conductivity (solute is a weak base and will partially ionize when dissolved)
Why are most solid ionic compounds electrically nonconductive, whereas aqueous solutions of ionic compounds are good conductors? Would you expect a liquid (molten) ionic compound to be electrically conductive or nonconductive? Explain.
Indicate the most important type of intermolecular attraction responsible for solvation in each of the following solutions:
(a) the solutions in [link]
(b) methanol, CH 3 OH, dissolved in ethanol, C 2 H 5 OH
(c) methane, CH 4 , dissolved in benzene, C 6 H 6
(d) the polar halocarbon CF 2 Cl 2 dissolved in the polar halocarbon CF 2 ClCFCl 2
(e) O 2 ( l ) in N 2 ( l )
(a) ion-dipole; (b) hydrogen bonds; (c) dispersion forces; (d) dipole-dipole attractions; (e) dispersion forces

Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?