| << Chapter < Page | Chapter >> Page > |
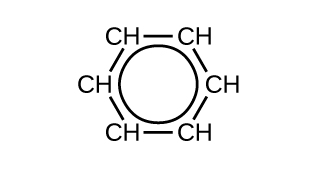
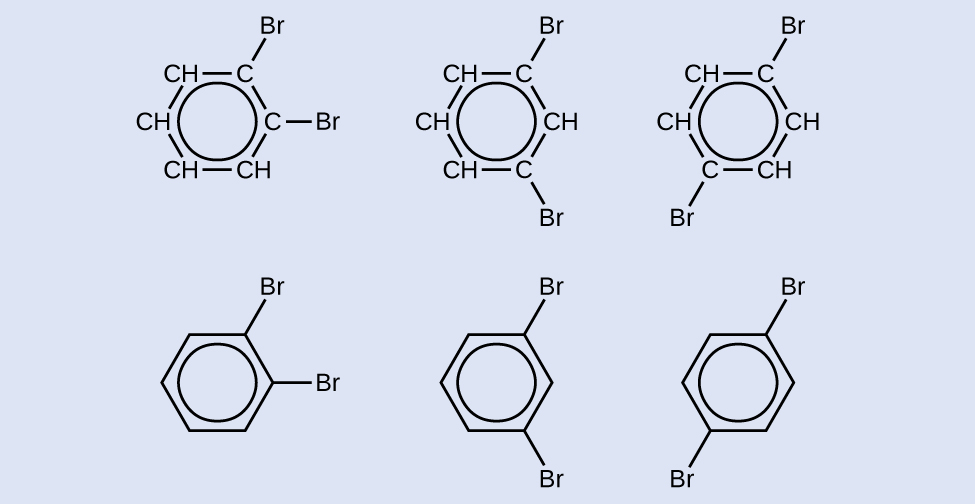
There are many derivatives of benzene. The hydrogen atoms can be replaced by many different substituents. Aromatic compounds more readily undergo substitution reactions than addition reactions; replacement of one of the hydrogen atoms with another substituent will leave the delocalized double bonds intact. The following are typical examples of substituted benzene derivatives:
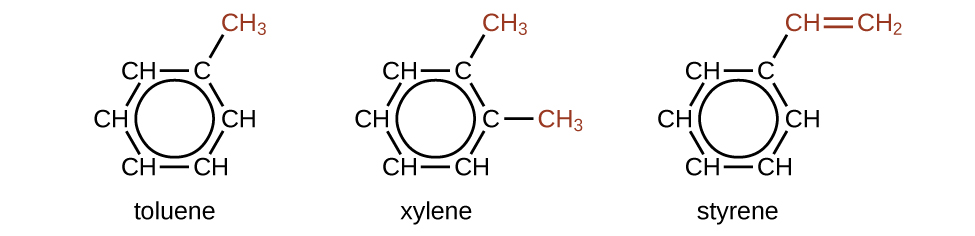
Toluene and xylene are important solvents and raw materials in the chemical industry. Styrene is used to produce the polymer polystyrene.
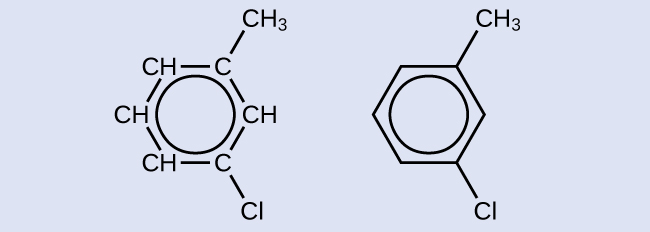
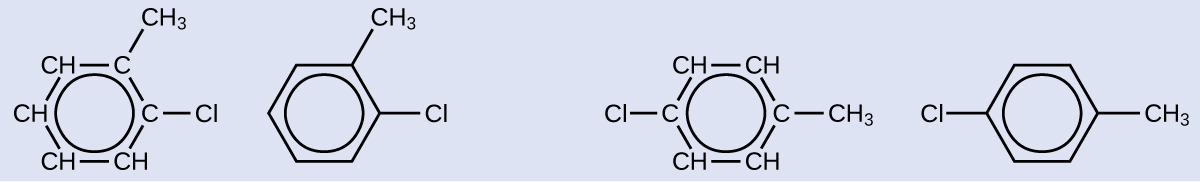
Strong, stable bonds between carbon atoms produce complex molecules containing chains, branches, and rings. The chemistry of these compounds is called organic chemistry. Hydrocarbons are organic compounds composed of only carbon and hydrogen. The alkanes are saturated hydrocarbons—that is, hydrocarbons that contain only single bonds. Alkenes contain one or more carbon-carbon double bonds. Alkynes contain one or more carbon-carbon triple bonds. Aromatic hydrocarbons contain ring structures with delocalized π electron systems.
Write the chemical formula and Lewis structure of the following, each of which contains five carbon atoms:
(a) an alkane
(b) an alkene
(c) an alkyne
There are several sets of answers; one is:
(a) C
5 H
12
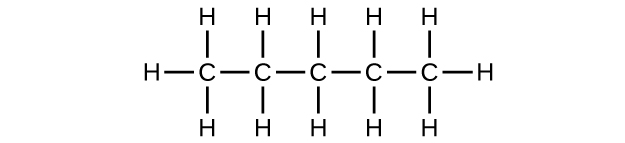 ;
;
(b) C
5 H
10
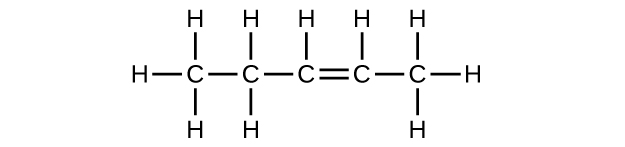 ;
;
(c) C
5 H
8
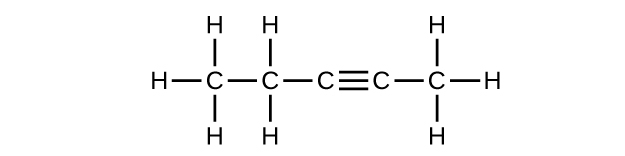
What is the difference between the hybridization of carbon atoms’ valence orbitals in saturated and unsaturated hydrocarbons?
On a microscopic level, how does the reaction of bromine with a saturated hydrocarbon differ from its reaction with an unsaturated hydrocarbon? How are they similar?
Both reactions result in bromine being incorporated into the structure of the product. The difference is the way in which that incorporation takes place. In the saturated hydrocarbon, an existing C–H bond is broken, and a bond between the C and the Br can then be formed. In the unsaturated hydrocarbon, the only bond broken in the hydrocarbon is the π bond whose electrons can be used to form a bond to one of the bromine atoms in Br 2 (the electrons from the Br–Br bond form the other C–Br bond on the other carbon that was part of the π bond in the starting unsaturated hydrocarbon).
On a microscopic level, how does the reaction of bromine with an alkene differ from its reaction with an alkyne? How are they similar?

Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?