| << Chapter < Page | Chapter >> Page > |
Electromagnetic radiation allows us to observe the world around us. Some materials and objects emit electromagnetic radiation and some reflect the electromagnetic radiation emitted by other objects (such as the Sun, a light bulb or a fire). When electromagnetic radiation comes from an object (whether the radiation is emitted or reflected by the object) and enters the eye, we see that object. Everything you see around you either emits or reflects electromagnetic radiation or both.
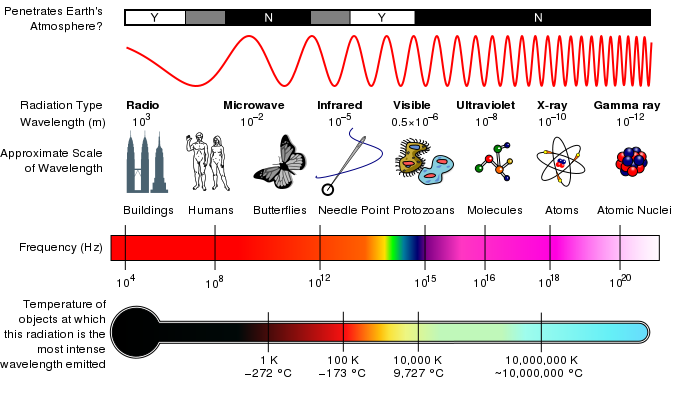
Electromagnetic radiation comes in a wide range of frequencies (or wavelengths) and the frequencies of radiation the human eye is sensitive to is only a very small part of it. The collection of all possible frequencies of electromagnetic radiation is called the electromagnetic spectrum, which (for convenience) is divided into sections (such as radio, microwave, infrared, visible, ultraviolet, X-rays and gamma-rays).
The electromagnetic spectrum is continuous (has no gaps) and infinite. In practice, we can only use electromagnetic radiation with wavelengths between (very roughly) (very high energy gamma rays) and (very long wavelength radio waves) due to technological limitations in the detectors used to receive electromagnetic radiation and in the devices used to produce or emit electromagnetic radiation.
The various frequencies (or wavelengths) of electromagnetic radiation coming from a particular object or material depends on how the object or material reflects and/or emits electromagnetic radiation.
[link] lists the wavelength- and frequency ranges of the divisions of the electromagnetic spectrum.
| Category | Range of Wavelengths (nm) | Range of Frequencies (Hz) |
| gamma rays | 1 | |
| X-rays | 1-10 | - |
| ultraviolet light | 10-400 | - |
| visible light | 400-700 | - |
| infrared | 700- | - |
| microwave | - | |
| radio waves |
Examples of some uses of electromagnetic waves are shown in [link] .
| Category | Uses |
| gamma rays | used to kill the bacteria in marshmallows and to sterilise medical equipment |
| X-rays | used to image bone structures |
| ultraviolet light | bees can see into the ultraviolet because flowers stand out more clearly at this frequency |
| visible light | used by humans to observe the world |
| infrared | night vision, heat sensors, laser metal cutting |
| microwave | microwave ovens, radar |
| radio waves | radio, television broadcasts |
When we talk of electromagnetic radiation as a particle, we refer to photons, which are packets of energy. The energy of the photon is related to the wavelength of electromagnetic radiation according to:
Planck's constant is a physical constant named after Max Planck.
J s
The energy of a photon can be calculated using the formula: or . Where E is the energy of the photon in joules (J), h is planck's constant, c is the speed of light, f is the frequency in hertz (Hz) and is the wavelength in metres (m).
Calculate the energy of a photon with a frequency of Hz
What is the energy of an ultraviolet photon with a wavelength of 200 nm?
We are required to calculate the energy associated with a photon of ultraviolet light with a wavelength of 200 nm.
We can use:

Notification Switch
Would you like to follow the 'Siyavula textbooks: grade 10 physical science [caps]' conversation and receive update notifications?